Solid Color & Gloss
spectro2guide
Color, Gloss and Haze Analysis of TiO2

"The first step is always the hardest." This also applies when formulating a coating. The application of the final finish determines the selection of the raw materials. Not only are mechanical properties like abrasion resistance and adhesion important, but also optical attributes like color, gloss and opacity; and one cannot forget the overall costs of the formulation. In order to guarantee consistent quality a routine quality control system needs to be established at the very first production step.
Paint is a liquid or powdery coating that is applied on objects as a very thin layer. By means of chemical or physical processes, it converts to an adherent film. Paint normally consists of the following components:
Titanium dioxide is the brightest, whitest pigment available. Due to its high refractive index (even higher than diamond) it effectively scatters the light and provides maximum opacity for a coating. Rutile is the most common natural form of TiO2 and preferred over anatase because of the lower photocatalytic activities and thus, better weather stability of the final coating.
The purity of TiO2 is process related. The chloride process makes up purer and brighter grades than the sulfate process. Additionally, impurities introduced by treatment chemicals or extraneous metal ions within the crystallites can degrade brightness. They usually discolor the pigment towards grey or yellow.
One way to perform a color measurement is to incorporate the TiO2 pigment into the coatings system of the final application. To ensure a smooth and homogeneous surface, the paint is applied to opacity drawdown charts with an automatic film applicator. The charts are made of black and white areas that are large enough to be measured with color instruments. An alternative is to take readings on dry pressed pucks. The pucks are made by applying high pressure to the dry TiO2 which is contained by a ring. The pressure is essential as it is the driving force to compact the pigment and hold it together for measurement. The face of the puck is then measured with a spectrophotometer.
The standardized CIE colorimetric values L* and b* are used to characterize brightness and undertone: the higher the L* value, the higher the brightness, the lower the b* value, the less yellow the appearance. In the table below, results for different grades of TiO2 are displayed. The spectro2guide can be used to measure CIELab color numbers. The instrument stores and directly transfers measurement data to smart-chart data analysis Software.
Besides brightness and undertone, optimum hiding power and tinting strength are properties that the TiO2 pigment has to meet.
| Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
|---|---|---|---|---|
| Brightness L* | 96.6 | 97.4 | 97.3 | 97.2 |
| Undertone b* | 2.1 | 1.5 | 1.5 | 1.5 |
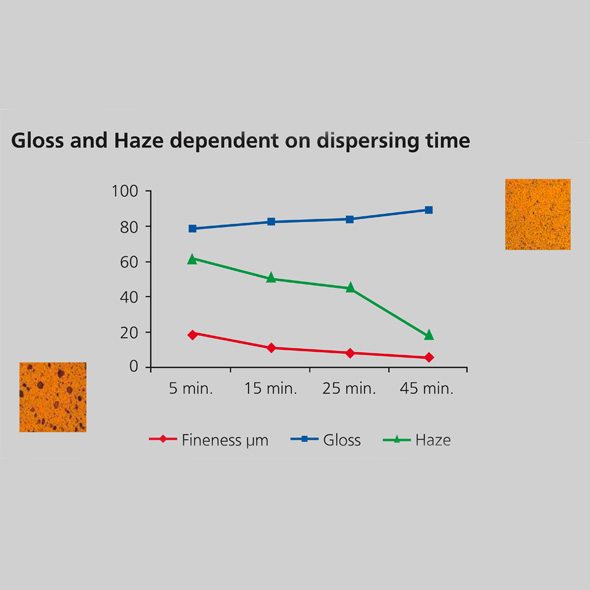
Gloss and haze level of a TiO2 pigment is mainly controlled by the primary particle size and the number of particles with a diameter larger than 0.5 μm. To achieve glossy finishes with improved distinctness of image, oversized particles have to be minimized. The haze-gloss is an objective tool to measure matte to high-gloss surfaces by offering three gloss geometries (20°, 60°, 85°) as well as reflectance haze measurement in one unit. However, to measure gloss and haze the TiO2 has to be incorporated in the coatings system and a drawdown has to be made.
During the dispersion process, the pigments are dispersed into small particles: the smaller the particles, the smoother the surface. The graph above shows the influence of degree of dispersion on gloss and haze. Pigment particles that are smaller than 10 μm show a tremendous reduction in haze and a slight increase in gloss, resulting in a glossy finish with improved imaging forming qualities.
Organic and inorganic absorption pigments selectively absorb and scatter the incident light. Besides the color itself, tinting strength is one of the most important properties that need to be controlled. Tinting strength is directly influenced by the pigment type and concentration used in the coating system.